The quality of evidence for each outcome was rated using the Grading of Recommendations Assessment, Development, and Evaluation (GRADE) system 30. If a study was evaluated as having a high risk of bias that might influence the pooled estimates of the outcome of interest, a subgroup analysis was conducted, excluding that study. All analyses were conducted in STATA® version 13.1 (StataCorp LP, College Station, Texas, USA). Number needed to treat (NNT) to avoid a particular adverse outcome was calculated as 1/(ACR × (1 − RR)), where ACR is the assumed control risk 26. An I 2 value of 0 per cent indicates no inconsistency between the results of individual trials, and an I 2 value of 100 per cent indicates maximal inconsistency. Heterogeneity was quantified with the I 2 measuring the proportion of variation (inconsistency) in the combined estimates 29. Statistical heterogeneity was examined as between‐study variation and tested using the Cochran Q test. Study design effect and unit of analysis was taken into consideration to calculate the standard error based on the original study design (a detailed explanation can be found in Appendix S1, supporting information) 26, 28. Accordingly, the studies were treated as random samples, assuming that the true effect varied between studies. The random‐effects model was chosen because the studies assessed different kinds of surgical incision and therefore clinical heterogeneity was expected. The meta‐analyses applied the random‐effects model of DerSimonian and Laird 27. The mean difference was used to summarize continuous data. 1).ĭata from each of the studies were organized in 2 × 2 contingency tables and used to calculate the relative risk (RR) for wound complications with associated 95 per cent c.i. 
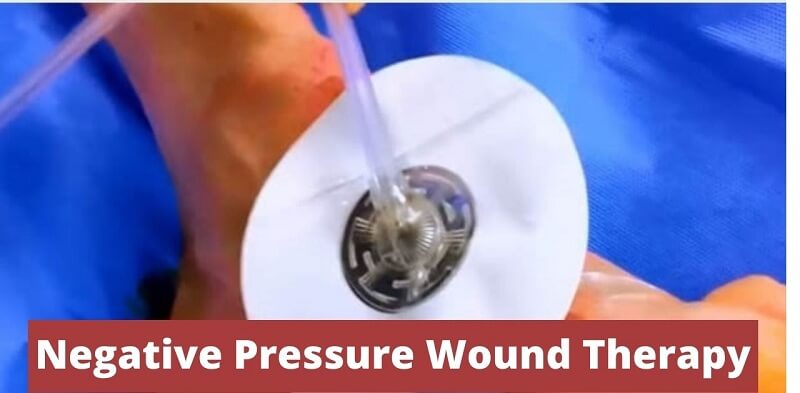
The mechanisms of action of this closed incision management have been supported by biomechanical studies: increased blood flow 14, 19 decreased lateral and shear stress at the suture lines with decreased risk of wound dehiscence 20 and increased lymph clearance with reduced formation of haematoma/seroma 21 ( Fig. In the latter case, the liquid is removed by evaporation through a semipermeable dressing. These NPWT devices consist of a single‐use battery‐powered negative‐pressure therapy device, an easy‐to‐place dressing, and either a very small and easily portable canister, or no canister at all. Two simplified NPWT devices became commercially available in 2010 (Prevena™ KCI) and 2011 (PICO™ Smith & Nephew, Hull, UK). Some of the first studies were case series 13, 14 and observational studies 15, 16 using one of the existing NPWT devices (VAC® KCI, San Antonio, Texas, USA) designed for open wounds 17, 18. In recent years, the indication for NPWT has been extended to include treatment of closed surgical incisions (incisional NPWT, iNPWT). The mechanisms of action include a characteristic pattern of blood flow around the wound, reduction in tissue oedema and stimulation of granulation tissue formation 10, 11, 12. The device ensures that negative pressure is transmitted to the wound bed and removes wound fluid 10. The dressing is then connected to a vacuum device via a drain or port. The wound is filled with a gauze or foam and sealed with an adhesive film dressing. Negative‐pressure wound therapy (NPWT) is usually used for the treatment of open wounds. Wound complications may also delay recovery, increase discomfort and reduce quality of life 8, 9. Postoperative wound complications may lead to increased healthcare costs due to prolonged inpatient stay, repeat surgery and the need for increased follow‐up 6, 7. Postoperative wound complications, such as infection, dehiscence, and formation of haematoma or seroma, are common complications of surgical procedures 1, 2, particularly among patients with risk factors such as obesity and diabetes 3, 4, 5.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
